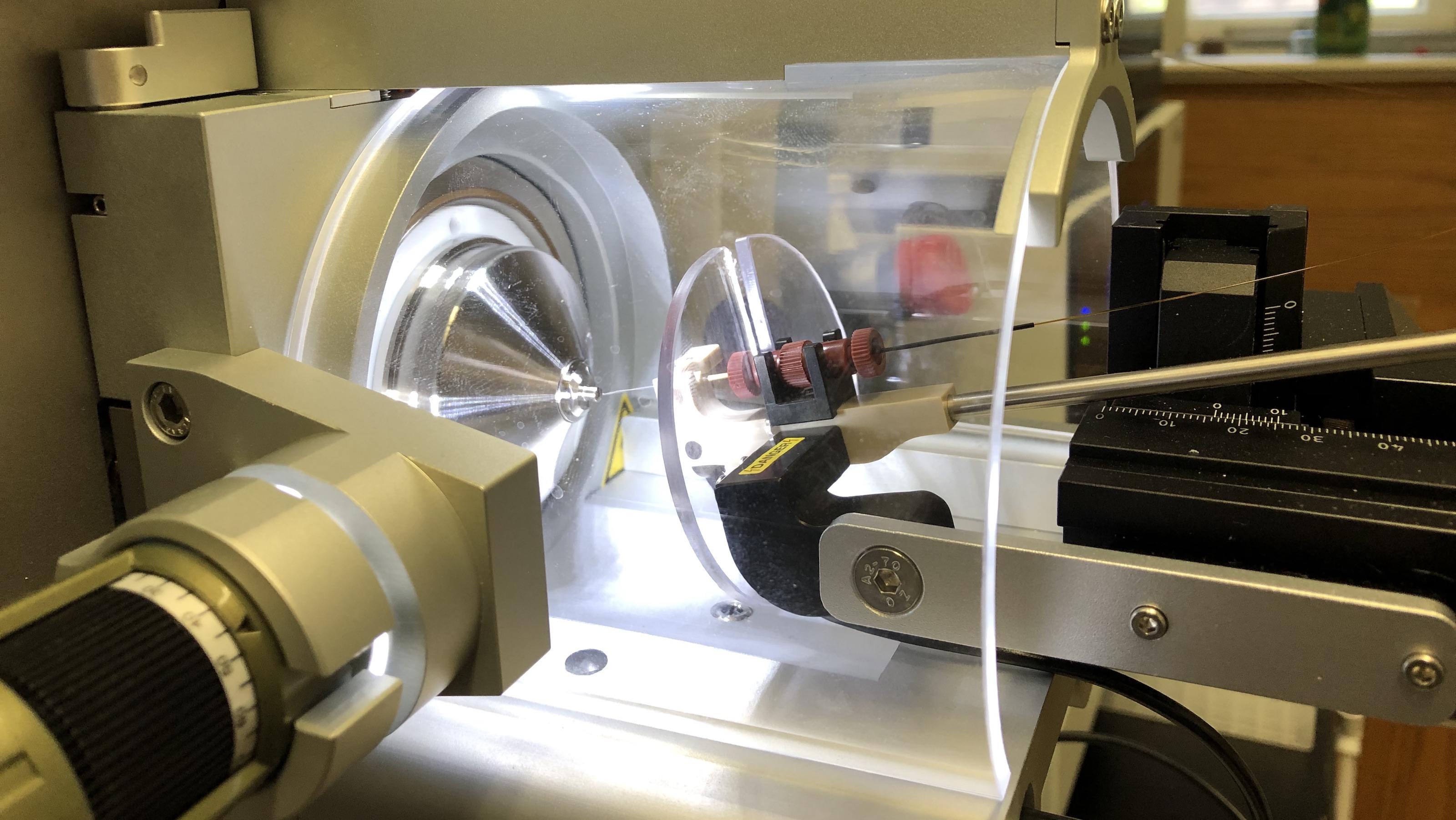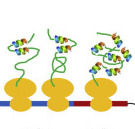
Protein Folding in Vivo
Co-translational folding;
Polysome dynamics;
Protein Folding systems & proteostasis
CROSSLINKING MASS SPEC
Method development;
Systems structural biology

LOOPABLE TRANSLATION
Teaching cells to make plastic-like proteins and to secrete them
PREBIOTIC PROTEINS
How did proteins learn to fold in the first place?